HI EVERYBODY!!
Welcome to Rainbow Creek. As most of the World is experiencing rainfall (or snowflakes) right now, I thought we might look into the old element of Water. The only thing wet and wild is You!
I am high and dry; seeing exactly No Rain from the Computer Cloud System. I am out on the creek watering my flowers with a hose while the weatherman says we have a 70% chance of rain! So your photostudy is on my rainbow creek.
Next, I went to the Google Index and typed in water. Many entries came up, but I selected the one from Wikipedia for our information study. You might want to check the other entries in the Google Index for yourself, just to see what is there.
Your Vid selections are from the Google You Tube Library and offer many points of view about water. Warning: You may want to make a trip to your bathroom before viewing the water vids! Enjoy my flowers on the creek!
https://en.wikipedia.org/wiki/Water
Water
From Wikipedia, the free encyclopedia
Water is a chemical compound with the chemical formula H2O. A water molecule contains oneoxygen and two hydrogen atoms connected by covalent bonds. Water is a liquid at standard ambient temperature and pressure, but it often co-exists on Earth with its solid state, ice, and gaseous state (water vapor or steam). Water also exists in a liquid crystal state near hydrophilic surfaces.[1][2]
Water covers 71% of the Earth's surface,[3] and is vital for all known forms of life.[4] On Earth, 96.5% of the planet's water is found in oceans, 1.7% in groundwater, 1.7% in glaciers and the ice caps of Antarctica and Greenland, a small fraction in other large water bodies, and 0.001% in the air asvapor, clouds (formed of solid and liquid water particles suspended in air), and precipitation.[5][6] Only 2.5% of the Earth's water is freshwater, and 98.8% of that water is in ice and groundwater. Less than 0.3% of all freshwater is in rivers, lakes, and the atmosphere, and an even smaller amount of the Earth's freshwater (0.003%) is contained within biological bodies and manufactured products.[5]
Water on Earth moves continually through the hydrological cycle of evaporation and transpiration(evapotranspiration), condensation, precipitation, and runoff, usually reaching the sea. Evaporation and transpiration contribute to the precipitation over land.
Safe drinking water is essential to humans and other lifeforms even though it provides no calories or organic nutrients. Access to safe drinking water has improved over the last decades in almost every part of the world, but approximately one billion people still lack access to safe water and over 2.5 billion lack access to adequate sanitation.[7] There is a clear correlation between access to safe water and GDP per capita.[8] However, some observers have estimated that by 2025 more than half of the world population will be facing water-based vulnerability.[9] A recent report (November 2009) suggests that by 2030, in some developing regions of the world, water demand will exceed supply by 50%.[10] Water plays an important role in the world economy, as it functions as a solventfor a wide variety of chemical substances and facilitates industrial cooling and transportation. Approximately 70% of the fresh water used by humans goes toagriculture.[11]

Water in three states: liquid, solid (ice), and (invisible)water vapor in the air. Clouds are accumulations of water droplets, condensed from vapor-saturated air.
Chemical and physical properties
Main articles: Properties of water, Water (data page), and Water model
Water is the chemical substance with chemical formula H2O: onemolecule of water has two hydrogen atoms covalently bonded to a single oxygen atom.
Water appears in nature in all three common states of matter (solid, liquid, and gas) and may take many different forms on Earth: water vapor and clouds in the sky; seawater in the oceans; icebergs in the polar oceans; glaciers and rivers in the mountains; and the liquid in aquifers in the ground.
The major chemical and physical properties of water are:
- Water is a liquid at standard temperature and pressure. It is tasteless and odorless. The intrinsic colour of water and ice is a very slight blue hue, although both appear colorless in small quantities. Water vapour is essentially invisible as a gas.[12]
- Water is transparent in the visible electromagnetic spectrum. Thusaquatic plants can live in water because sunlight can reach them. Infrared light is strongly absorbed by the hydrogen-oxygen or OH bonds.
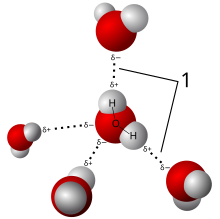
- Since the water molecule is not linear and the oxygen atom has a higher electronegativity than hydrogen atoms, it carries a slight negative charge, whereas the hydrogen atoms are slightly positive. As a result, water is a polar molecule with an electrical dipole moment. Water also can form an unusually large number of intermolecular hydrogen bonds (four) for a molecule of its size. These factors lead to strong attractive forces between molecules of water, giving rise to water's high surface tension[13] and capillary forces. The capillary action refers to the tendency of water to move up a narrow tube against the force of gravity. This property is relied upon by all vascular plants, such as trees.[14]
- Water is a good polar solvent and is often referred to as the universal solvent. Substances that dissolve in water, e.g., salts, sugars, acids, alkalis, and some gases – especially oxygen, carbon dioxide (carbonation) are known as hydrophilic (water-loving) substances, while those that are immiscible with water (e.g., fats and oils), are known as hydrophobic (water-fearing) substances.
- Most of the major components in cells (proteins, DNA and polysaccharides) are also dissolved in water.
- Pure water has a low electrical conductivity, but this increases with the dissolution of a small amount of ionic material such as sodium chloride.
- The boiling point of water (and all other liquids) is dependent on the barometric pressure. For example, on the top of Mt. Everest water boils at 68 °C (154 °F), compared to 100 °C (212 °F) at sea level. Conversely, water deep in the ocean near geothermal vents can reach temperatures of hundreds of degrees and remain liquid.
- At 4181.3 J/(kg·K), water has a high specific heat capacity, as well as a high heat of vaporization(40.65 kJ·mol−1), both of which are a result of the extensive hydrogen bonding between its molecules. These two unusual properties allow water to moderate Earth's climate by buffering large fluctuations in temperature.
- The maximum density of water occurs at 3.98 °C (39.16 °F).[15] It has the anomalous property of becoming less dense, not more, when it is cooled to its solid form, ice. During freezing, the 'open structure' of ice is gradually broken and molecules enter cavities in ice-like structure of low temperature water. There are two competing effects: 1) Increasing volume of normal liquid and 2) Decrease overall volume of the liquid. Between 0 and 3.98 °C, the second effect will cancel off the first effect so the net effect is shrinkage of volume with increasing temperature.[16] It expands to occupy 9% greater volume in this solid state, which accounts for the fact of ice floating on liquid water, as in icebergs.
- The density of liquid water is 1,000 kg/m3 (62.43 lb/cu ft) at 4 °C. Ice has a density of 917 kg/m3 (57.25 lb/cu ft).
- Water is miscible with many liquids, such as ethanol, in all proportions, forming a single homogeneous liquid. On the other hand, water and most oils are immiscible, usually forming layers according to increasing density from the top. As a gas, water vapor is completely miscible with air.
- Water forms an azeotrope with many other solvents.
- Water can be split by electrolysis into hydrogen and oxygen.
- As an oxide of hydrogen, water is formed when hydrogen or hydrogen-containing compounds burn or react with oxygen or oxygen-containing compounds. Water is not a fuel, it is an end-product of the combustion of hydrogen. The energyrequired to split water into hydrogen and oxygen by electrolysis or any other means is greater than the energy that can be collected when the hydrogen and oxygen recombine.[17]
- Elements which are more electropositive than hydrogen such as lithium, sodium, calcium, potassium andcaesium displace hydrogen from water, forming hydroxides. Being a flammable gas, the hydrogen given off is dangerous and the reaction of water with the more electropositive of these elements may be violently explosive.
See also
- The water (data page) is a collection of the chemical and physical properties of water.
Water is described in many terms and contexts:
- according to state
- solid – ice
- liquid – water
- gaseous – water vapor
- plasma
- according to meteorology:
precipitation according to movement precipitation according to state - vertical (falling) precipitation
- rain
- freezing rain
- drizzle
- freezing drizzle
- snow
- snow pellets
- snow grains
- ice pellets
- frozen rain
- hail
- ice crystals
- horizontal (seated) precipitation
- liquid precipitation
- solid precipitation
- mixed precipitation
- in temperatures around 0 °C
- vertical (falling) precipitation
- according to occurrence
- groundwater
- meltwater
- meteoric water
- connate water
- fresh water
- surface water
- mineral water – contains many minerals
- brackish water
- dead water – strange phenomenon which can occur when a layer of fresh or brackish water rests on top of denser salt water, without the two layers mixing. It is dangerous for ship traveling.
- seawater
- brine
- according to uses
- tap water
- bottled water
- drinking water or potable water – useful for everyday drinking, without fouling, it contains balanced minerals that are not harmful to health (see below)
- purified water, laboratory-grade, analytical-grade or reagent-grade water – water which has been highly purified for specific uses in science or engineering. Often broadly classified as Type I, Type II, or Type III, this category of water includes, but is not limited to, the following:
- according to other features
- soft water – contains fewer minerals
- hard water – from underground, contains more minerals
- distilled water, double distilled water, deionized water – contains no minerals
- water of crystallization — water incorporated into crystalline structures
- hydrates — water bound into other chemical substances
- heavy water – made from heavy atoms of hydrogen – deuterium. It is in nature in normal water in very low concentration. It was used in construction of first nuclear reactors.
- tritiated water
- according to microbiology
- according to religion
Other topics
- Dihydrogen monoxide hoax
- Hydrational fluids
- Mirage
- Mpemba effect
- Water Pasteurization Indicator
- Water pinch analysis
https://en.wikipedia.org/wiki/Water
http://www.youtube.com/watch?v=nSENolWbyYQ
http://www.youtube.com/watch?v=8nIaQWRBQ6E
...this is brendasue signing off from
Rainbow Creek.
See You next time! Sending Joy out for You!
(Thanks everybody for stopping by and all the +1s and comments. I hope You are learning some stuff!)
Of course, one more great performance:
http://www.youtube.com/watch?v=YLcURLyP6WM
O+O

























No comments:
Post a Comment
Hi Everybody! Please say hello and follow so I know you are here! Due to the inconsideration of people trying to put commercials on my blog comment area, I have restricted use of anonymous posts. Sorry that some hurt all.
My public email is katescabin@gmail.com No spammers or trolls